Electron Configuration
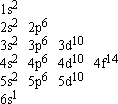
This is the electron configuration for Gold. To have a stable nucleus gold has 79 electrons organized in the diagram to the left.
Valance Electrons, Quantum Numbers and Bohr Model Diagram
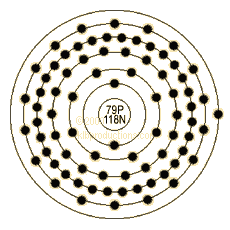
Valance electrons are the electrons on the outermost ring. They can react with other elements during chemical reactions. Gold's dot diagram to the left contains one valance electron.
The four quantum numbers for the last electron in gold's orbital diagram are 5,2, 2, -1/2.
This is also the Bohr Model Diagram.
The four quantum numbers for the last electron in gold's orbital diagram are 5,2, 2, -1/2.
This is also the Bohr Model Diagram.
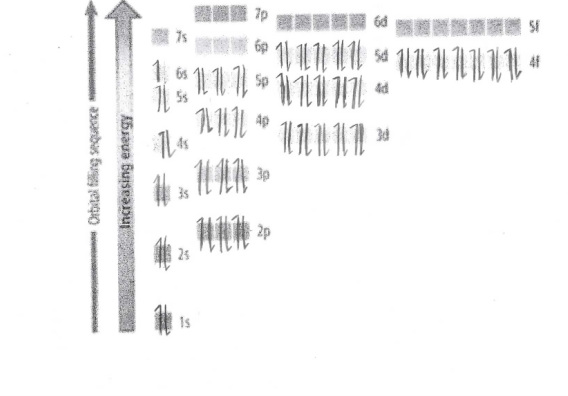
This is the Energy Diagram for Gold: